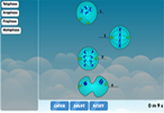
Atom diagram labelling game
Atoms of different elements are the basic units that form matter – from the particles of air you breathe, to your favourite food or clothes.
Dive Deep into the World of Atoms: Label and Learn
Atoms are the tiny building blocks of everything we see, touch, and interact with daily. To truly grasp the intricacies of our universe, understanding the atom and its components is crucial. Through engaging, interactive diagrams and games, children can not only learn about atoms but also understand their importance in the grand scheme of things. Let's explore the captivating journey of labelling parts of an atom.
Atom Diagram: The Blueprint of Matter
Before diving into the parts, it's essential to understand what an atom diagram represents. Just as a map provides guidance about a city or country, an atom diagram gives a visual representation of the structure of an atom. It shows the central nucleus and the orbiting electrons, laying down the basics of atomic structure.
Diagram of Atoms Labelling: Deciphering the Components
The diagram of atoms labelling is a step-by-step guide to identifying the different parts of the atom. Here's a brief rundown:
Nucleus: Located at the center of the atom, the nucleus houses protons (positively charged) and neutrons (no charge).
Electrons: These negatively charged particles revolve around the nucleus in specific orbits or shells.
Protons: Positive particles within the nucleus.
Neutrons: Neutral particles, also within the nucleus.
Understanding and identifying these parts is crucial for a comprehensive grasp of atomic theory, which underpins so many scientific concepts.
Label Parts of an Atom: Interactive Learning
One of the most effective ways to reinforce knowledge about atomic structure is through interactive games where learners can label parts of an atom. By actively engaging with the material and labeling various components, kids can better retain and recall information.
For those eager to dive into this interactive experience, the atom diagram labelling game is an excellent place to start. This tool combines the thrill of a game with essential educational content, ensuring kids grasp the basics while having fun.
Expanding Horizons with Science Games
Science is not just about rote learning. It's about exploration, inquiry, and hands-on understanding. Integrating games into the learning process makes this journey even more compelling. For a wide range of interactive educational experiences, explore the array of science games here. Whether it's biology, chemistry, or physics, there's something for every budding scientist.
Conclusion: The Atom - A Universe in Itself
The journey into the world of atoms is both fascinating and foundational. Through the diagram of an atom and the active process of labelling, children gain a deeper appreciation for the micro-universe that exists in every speck of matter. Dive into this atomic adventure, and discover the wonders that lie within the smallest of structures.
While the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom.
In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. You can compare such distances to how many millions of kilometers each planet is to our Sun.
While an atom is usually described as the building block of all matter, it does have three subatomic components. These are the protons, neutrons, and charged clouds called electrons. The neutrons and protons are all clumped together to form the nucleus.
The number of protons possessed by an atom determines which element it is. Normally, there are as many protons as there are electrons, meaning that their positive and negative charges cancel out. When electrons are either added or lost, the atom is ionized.
Your kids or students can try our atom diagram labelling to test their knowledge on the parts of an atom and aid in the assessment of their comprehension of basic chemistry principles.
In the rapidly evolving landscape of K-12 education, the integration of digital tools has become indispensable. Welcome to your ultimate destination for mastering Atom Diagram Labelling. Whether you are a dedicated educator seeking to invigorate your classroom dynamics or a parent aiming to support your child's learning journey at home, this page offers a curated selection of resources designed to deliver results. Our platform specializes in gamified learning, a method proven to increase student engagement and retention rates significantly.
This comprehensive guide explores the multifaceted aspects of draw and label the parts of an atom and related concepts. We have consolidated a vast array of keywords and topics—from fundamental definitions to advanced applications—to ensure you have everything you need in one place. By leveraging the power of interactive play, we transform complex educational standards into accessible, enjoyable experiences for students of all abilities.
The Educational Value of Gamifying Atom Diagram Labelling
Why choose games over traditional rote memorization? Research consistently demonstrates that interactive learning facilitates deeper cognitive processing. When students engage with our Atom Diagram Labelling games, they are not merely passive recipients of information; they are active participants in their own education.
Cognitive Benefits
- Active Recall: Games that require quick thinking force the brain to retrieve information rapidly, strengthening neural pathways associated with Atom Diagram Labelling.
- Immediate Feedback: Unlike paper worksheets, our digital activities provide instant validation. Students know immediately if they have understood diagram label the parts of an atom, allowing for real-time correction and learning.
- Motivation and Engagement: The competitive element of games—earning points, beating high scores—drives intrinsic motivation. Students practice longer and more frequently when the work feels like play.
Core Concepts and Vocabulary
To achieve proficiency in Atom Diagram Labelling, a strong command of specific vocabulary is essential. Below, we break down the key terms you will encounter in our activities. Mastering these will provide a solid foundation for more advanced study.
- Draw And Label The Parts Of An Atom: An essential component of the curriculum. Understanding draw and label the parts of an atom allows students to tackle more complex problems and communicate their ideas effectively.
- Diagram Label The Parts Of An Atom: An essential component of the curriculum. Understanding diagram label the parts of an atom allows students to tackle more complex problems and communicate their ideas effectively.
- Diagram Of An Atom Labeled: An essential component of the curriculum. Understanding diagram of an atom labeled allows students to tackle more complex problems and communicate their ideas effectively.
- Labelled Diagram Of Atom: An essential component of the curriculum. Understanding labelled diagram of atom allows students to tackle more complex problems and communicate their ideas effectively.
- Labeled Atom Diagram: An essential component of the curriculum. Understanding labeled atom diagram allows students to tackle more complex problems and communicate their ideas effectively.
- Atom Diagram Labeled: An essential component of the curriculum. Understanding atom diagram labeled allows students to tackle more complex problems and communicate their ideas effectively.
- Label The Atom: An essential component of the curriculum. Understanding label the atom allows students to tackle more complex problems and communicate their ideas effectively.
- Atom Labeling: An essential component of the curriculum. Understanding atom labeling allows students to tackle more complex problems and communicate their ideas effectively.
By regularly exposing students to these terms in varied contexts—such as Atom Diagram Labelling memory games, crossword puzzles, and interactive quizzes—educators can ensure that vocabulary moves from short-term memory to long-term retention.
How to Integrate These Resources into Your Curriculum
Flexibility is the hallmark of modern teaching aids. Here is a step-by-step guide on how to effectively utilize our Atom Diagram Labelling resources in different educational settings.
For Classroom Teachers
Warm-Up Activity: Start your class with a 5-minute session of atom labeled. It acts as an excellent icebreaker to get students focused.
Guided Practice: Use our interactive diagrams or quizzes on a smartboard. Call on students to solve problems collectively, fostering group discussion and peer learning.
Assessment: Assign specific games as homework or exit tickets. The instant scoring allows you to track progress without hours of grading.
For Parents and Homeschoolers
Create a structured routine where learning feels like a reward. After completing a traditional lesson on Atom Diagram Labelling, allow your child to 'play' for 15 minutes. This reinforces the lesson immediately. Encourage them to explain the game mechanics to you; teaching back is one of the most effective ways to solidify knowledge.
Frequently Asked Questions about Atom Diagram Labelling
How can I improve my skills in Atom Diagram Labelling?
Consistency is key. We recommend setting aside 10-15 minutes daily to engage with our draw and label the parts of an atom activities. Spaced repetition is highly effective for mastering new material.
Are these resources suitable for all ages?
Yes, while designed with K-12 students in mind, our resources for Atom Diagram Labelling are scalable. Beginners can start with basic vocabulary matching, while advanced learners can tackle complex logic puzzles and grammar challenges.
Explore Related Topics
Learning is a connected journey. Once you have mastered Atom Diagram Labelling, we encourage you to explore these related areas to broaden your understanding:
atom to label, atom labeled.
Our extensive library ensures that whether you are looking for remedial practice or enrichment activities, you will find exactly what you need.
Conclusion
Thank you for choosing us as your trusted partner in education. We are committed to providing high-quality, free access to the best Atom Diagram Labelling games and worksheets available online. Bookmark this page, share it with your colleagues and friends, and return often—we are constantly updating our database with new activities to keep learning fresh and exciting. Start exploring draw and label the parts of an atom today and watch your students thrive!